|
To find a story that provides a thumbnail illustration for the nature and progress of worldly science, one needs to look no further than the medicine cabinet of any home. Chances are it contains aspirin. If not, then look for one of aspirin’s many cousins, pain killers that, despite their long and widespread usages, never cease to make headline news.
After eons of prehistory as folk remedy, aspirin emerged in 1899 in one of the world’s first industrial research laboratories. Aspirin has many therapeutic effects. At over-the-counter dosage (one or two grams), it relieves fever and minor aches and pains. At dosages three or four times higher, available by prescription only, it reduces swelling and is used to treat gout, rheumatoid arthritis, and inflammatory ailments.[i] Many people take low dosages (below 100 milligrams) daily for preventing recurrent stroke or heart attack.[ii] Recent studies found it effective in reducing risks for colon and breast cancers.[iii] Evidence is accumulating for similar effects in Alzheimer and other diseases.[iv]
A great complaint against aspirin is that it irritates the stomach and in some cases cause ulcer and internal bleeding. Despite these and other undesirable side effects, it is widely used. It made the 1950 Guinness Book of Records as the world’s most popular pain-killing drug. Now more than 80 million tablets of it are being consumed every day in the United States alone, and for many more purposes.
Success breeds competition. Some fifty cousins of aspirin exist, most of which appeared after World War II. Together they constitute a family known as non-steroidal anti-inflammatory drugs (NSAIDs). Popular over-the-counter NSAIDs include aspirin (Bayer, Ecotrin), ibuprofen (Advil, Motrin), and naproxen (Aleve).[v] Many more are available by prescription. They have different chemical structures. Yet they all suppress pain, fever, and inflammation and irritate the stomach to various degrees.
A new lineage nicknamed “super aspirin” appeared at the end of the twentieth century. Coxib (Celebrex, Vioxx) achieves similar therapeutic profiles as the old NSAIDs but is gentler on the stomach. These drugs are rationally designed based on scientific knowledge, but alas, they have their own shortfalls.[vi]
While consumers are happy that aspirin works so well in so many areas, scientists are excited in understanding how it works and finding ways to make it work better. They have come a long way since the 1970s and realize that many more secrets await discovery. Aspirin itself is a small chemical molecule, the properties of which have been known for more than a century. However, the living body with which it must interact as a medicinal agent is most complex and not well understood despite scientific advancement. Aspirin research involves many approaches that will be discussed in more details later in the book: cut and try, educated guess, breakthroughs and setbacks, laboratory experiments, theories and controversies, synthesis of knowledge from many disciplines, clinical trials with definitive or inconclusive results, and judgments based on incomplete knowledge.
Contrary to the stereotype of applied science as perfunctory deployment of complete knowledge swayed only by cultural tastes, aspirin tells how judicious applications themselves demand and generate new scientific knowledge. Its story reveals worldly science as a trustworthy friend in the tough journey of life. As you make your way through the knotty problems of the world, you develop a deeper understanding of your friend, the world, and perhaps yourself, so that you are better prepared for the new problems to come.
From folk remedy to pure chemical
Aspirin is the generic medical name for the chemical acetylsalicylic acid, a derivative of salicylic acid. Compounds of salicylic acid are found in some plants, notably white willow and meadowsweet (Spirea ulmaria). Acetyl- and spirea inspired the name aspirin.[vii]
The medicinal effects of the plants were discovered by people through life experiences. An Egyptian papyrus prescribed willow leaves for fever and swelling. The Greek Hippocrates, who was honored as the father of medicine, recommended willow bark for pain and fever. Medieval medical scripts contained many references to the plant.
Willow bark made its way into scientific literature in 1763 through the work of English vicar Edmund Stone. It had been among folk remedies of rural workers. Stone decided to pick it out for study because he believed that remedies came in the vicinity of ailments and observed that both willows and fevers occurred commonly in swampy areas. He dispensed a fixed amount of willow bark powder every four hours to many feverish patients, recorded the results, and wrote a letter complete with a speculative explanation of its efficacy to the Royal Society of London. The specific explanation he gave turned out to be wrong and his clinical method was crude. Nevertheless, his systematic approach and attempt at a principled explanation were close to that of science.
Chemistry was fast transforming from alchemy into a natural science even as Stone dispensed willow bark. One of its first applications was in drug preparation. Hitherto the medicinal repertoire consisted of herbs and other natural products. Soon pharmacologists used their knowledge of chemical analysis to isolate and purify “active principles,” for instance to isolate morphine from opium.
Isolation processes may destroy the complexity by which several ingredients in a natural product work in concert to produce the observed therapeutic effects, for example by subtly neutralizing some side effects of isolated active principle. Nevertheless, in many ways purified extracts are superior to herbal medicine. They are easier to take, more uniform in quality, and allow more precise therapeutic dosages. Equally important, their convenience in storage and transportation make them more affordable.
The advantages of extracts extend beyond besides into laboratories. They enabled researchers to experiment with the drugs, study their interactions with other chemicals and with human physiology, measure the effects of dosage variation, modify them to make new chemicals, and find alternative compounds for better therapeutic results. Thus pharmacology, although practical in its major intent, repays its debt to natural science by contributing to the advancement of chemical knowledge.
In 1828, German pharmacologists isolated from willow bark a yellow bitter crystal, which they called salicin. Swiss pharmacologists isolated a similar substance from meadowsweet. Ten years later, French chemists synthesized salicylic acid. Physicians administered the two compounds to patients, whose symptoms they observed and whose urine they analyzed. They found that taking both compounds reduced rheumatic fever, and salicin was transformed in the body to salicylic acid. Based on the observations, they identified salicylic acid as the active medicinal ingredient responsible for willow bark’s efficacy in relieving pain and fever.
Scientists extract or synthesize spoonfuls of a chemical and are happy that it suffices for laboratory experiments. As a useful drug for the mass of people, however, large quantity of the stuff at reasonable prices is required. To develop cost effective processes for mass production, science and technology again come into play.
Soon after chemical extracts and synthetics appeared, they were in demand. Supply of drugs came from two kinds of companies, pharmacies and chemical manufacturers. Some pharmacies, which traditionally prepared the portions prescribed by physicians, set up laboratories and factories and morphed into pharmaceutical firms. Among them was Merck, an early seller of salicin. As foundation of the modern science-based pharmaceutical industry, however, pharmacies were perhaps less important than manufacturers of fine chemicals – to this day the U.S. national accounting classifies pharmaceuticals under “chemicals and allied products.”[viii]
The first fine chemicals to have wide social impacts were dyes, which brightened life by turning drab garments into a rainbow of colors. The dyes industry grew up alongside organic chemistry in the second half of the nineteenth century. Their mutual stimulations exemplified the coupled dynamics of science and business. Dyes are complex organic chemicals. Much technical knowledge is required to find dyes for desirable colors, make them adhere to popular fabrics, and ensure their color-fastness. To meet these challenges the German firms that manufactured dyes from coal tar initiated organizational innovations of lasting impact. They invented industrial research laboratories and forged close ties with research universities, which emerged around the same time. These institutions contribute crucially to the enabling environment in which worldly sciences thrive.[ix]
Anyone who has tried to wash stained hands knows the affinity of dyes to living tissues. Scientists went one step further and put the knowledge to use. Since 1849, they had been using dyes to stain otherwise colorless biological specimen and make them visible under the microscope. Noting the different susceptibilities of various cells and bacteria to various dyes, they developed differential staining criteria to identify and classify types of cell, for example, a particular staining differentiates two kinds of bacteria with different cell walls. Toward the end of the century Paul Ehrlich and other chemists suggested that staining was a chemical reaction between a dye and a bacterium or a cell. Because cells were susceptible to specific staining, perhaps dyes may be harnessed for chemotheurapeutic purposes.[x] Such ideas were not lost on the dyes firms. As the market for dyes matured, those looking to diversify turned their scientific and organization prowess to drug development.
Bayer was one of three leading research-intensive dyes firms. Its research supervisor Carl Duisberg, a chemist who later became the firm’s chief executive, contributed much to instituting the organization of modern industrial research. He directed company researchers into the drug area, and after initial sucesses set up in 1896 an independent drug laboratory. It had a pharmaceutical division for drug discovery, directed by Arthur Eichengrün, and a pharmacological division for testing the drugs, directed by Heinrich Dreser.
Among Eichengrün’s first ideas was to find a derivative of salicylic acid that would be as therapeutically effective but with less undesirable side effects. He hired a young chemist Felix Hoffmann who, because of his arthritic father, had a personal passion in the project. Hoffmann came up with acetylsalicylic acid.[xi]
At first acetylsalicylic acid failed even to win everyone at Bayer. Eichengrün tested it on himself and pushed it vigorously. Dreser thought it was just a better-tasting salicylic acid unworthy of production. As the two heads of research quarreled, acetylsalicylic acid languished on the shelf. Finally Duisberg stepped in and had it tested by outside pharmacologists and physicians. They brimmed with enthusiasm. Dreser changed his mind and published a paper that did not mention Eichengrün and Hoffmann. Bayer launched acetylsalicylic acid as a commercial drug in 1899 under the name Aspirin.
Research, development, marketing
Three innovations of the dyes industry became invaluable legacies of the pharmaceutical industry. The first was research and development, of which aspirin was among the first pharmaceutical products. Emphasis on science and research remains a distinctive tradition of the pharmaceutical industry.
The second innovation was in marketing. The dyes industry pioneered the practice of targeting not ordinary consumers but professionals. It hired technical salesmen to explain to other technicians how to use various dyes on various fashionable fabrics. Adapting this approach, Bayer mounted an extensive sales campaign for aspirin targeting physicians. It initiated the drug marketing strategy “to help doctors to help their patients,” which thrives alongside advertisements prompting consumers to “ask your doctor about it.”[xii]
The dyes industry was also in the forefront in asserting intellectual property rights. Favorable patent laws play important roles in the pharmaceutical and life science industries; witness the recent scramble to patent human genes.[xiii] However, they did not benefit aspirin. Bayer settled for registering a trademark for the name Aspirin.[xiv] It did not patent acetylsalicylic acid, not because it would not but because it could not. The chemical was old stuff, synthesized by French chemist Charles Frederic Gerhardt back in 1853.
The significance of Bayer’s work on aspirin lies not in the discovery of a chemical compound but in the development of a compound into a useful drug. Turning a chemical into a drug calls for extensive research to identify its potential applications and markets, evaluate its clinical effects, optimize its properties, and design efficient manufacturing processes. The research generates application related knowledge that marks the difference between discovering something and discovering it as something useful, or between pure and worldly sciences.
To discover what a thing is good for requires knowledge about relevant situations, which are often subtle and difficult. Lack of such knowledge partly explains why many chemicals sat on the shelf for decades before their therapeutic values were realized. This happened to aspirin’s rival Tylenol. Its active ingredient was synthesized in 1878, but had to wait until 1955 before being developed into a popular drug.[xv] Ever more revealing are the stories of antibacterial drugs. Sulfanilamide was synthesized in 1908, but it was the discovery of its therapeutic effectiveness in 1932 that won a Nobel Prize. Penicillin was discovered in 1928 and its therapeutic properties in 1939, and both discoveries were cited in the Nobel Prize. In Nobel Prizes such as these, the scientific community acknowledges the equal scientific importance of discovering and developing a drug. Unfortunately, this point is often overlooked in science studies, so that Hoffmann is often accorded with the credit for aspirin, to the neglect of Eichengrün and others in Bayer.[xvi]
The importance of analytic techniques
Aspirin works. Willow bark works; it is available as herbal medicine and clinical trials find it effective against osteoarthritic pain.[xvii] The old NSAIDs work. Chemical analysis brought more knowledge about what are working – chemical molecules whose structures are known precisely. However, for seven decades physiological and pharmacological knowledge of the old NSAIDs did not advance much beyond that of willow bark. Both remained on the empirical and phenomenological level. People knew from experience that aspirin worked, but lacked scientific knowledge about how it worked and why it worked like its cousins. New York Times Magazine aptly called it in 1966: “The wonder drug nobody understands.”[xviii]
Discovering mechanisms underlying disparate phenomena is the font of basic science. Science, especially biochemistry and molecular biology, advanced tremendously since aspirin made its debut. The castle of NSAIDs’ working principles was still intact, but siege engines were ready. The first to breach the wall in 1971, and would receive a Nobel Prize for it, was pharmacologist John Vane.[xix]
No scientific discovery is solely the work of a single person, as acknowledged in Isaac Newton’s famous aphorism: “If I have seen further, it is by standing on ye shoulders of giants.” So it was with Vane. He was brought to aspirin in 1968 by Henry Collier, a pharmacologist who had worked on it for a decade. Collier had discovered that although both morphine and aspirin kill pain, they act by different principles. Morphine acts on the brain. Aspirin acts locally at the sites of injury. What local biochemical mechanisms underlie aspirin’s actions? Collier’s research was stymied, partly because his tools and techniques were rather blunt.
Collier experimented with whole animals such as guinea pigs and rabbits. He injected an animal with a pathology-inducing chemical and then a drug, observed the animal’s responses and analyzed its blood and tissues. By varying the pathogen and the drug, he hoped to tease out what acted on what and how. After numerous experiments, no pattern emerged. An animal’s body harbors millions of chemicals and hundreds of biochemical pathways. It is so complex a medium that therapeutic mechanisms are easily obscured. Furthermore, biopsy and blood analysis, which take time to perform, may not be able to capture fleeting biochemical reactions. Frustrated, Collier turned to Vane, an expert in bioassay. The relative successes of the two scientists illustrate the importance of experimental techniques and instruments in research.
Vane’s method depends on the central tenet of biochemistry and molecular biology: What occur inside a living body are not mysterious “vital forces.” Rather, they are chemical and physical processes that obey the laws of chemistry and physics and can also occur outside the body, in tissue culture, cell culture, or even cell-free medium. Cells in a dish or molecules in a test tube are much simpler and easier to manipulate than living organisms, hence much more susceptible to analysis and understanding. This partly explains the increasing importance of biochemistry and molecular biology in biology.
Over the years, researchers have assembled a large library of how a kind of tissue reacts physically and chemically to various kinds of irritants. For instance, a tissue secrets a specific substance when it is exposed to a chemical known to cause inflammation in people, and that substance in turn causes another tissue to twitch. A bioassay test exposes a piece of partially known tissue to a novel environment and records the tissue’s reaction to figure out unknown characteristics of it or the environment. Vane had developed a powerful bioassay technique in which a sequence of tissues probed a chain of chemical reactions. When Collier approached him, he agreed to investigate what happened when he exposed tissues to pain-inducing chemicals, and what happened if he added aspirin to the chemicals.
With bioassay experiments, Vane soon found that aspirin inhibits the production of a mysterious substance. Without aspirin, that substance would be produced by guinea pig lungs in shock and caused rabbit aorta to contract. What is it that aspirin preempted? Vane took eighteen months to identify it as a prostaglandin. Many experiments by him and his group confirmed his conjecture: aspirin inhibits the production of prostaglandins.[xx]
What are prostaglandins, the production of which aspirin inhibits? They constitute a class of unsaturated fatty acids produced by cells in many parts of the body. Discovered in the 1920s, they excited much scientific excitement in the late 1960s.
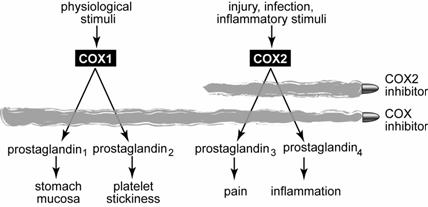
Many kinds of prostaglandin exist in the body to serve a plethora of physiological functions, some of which are irritable, others beneficial. Prostaglandins are among the chemicals secreted by the body’s immune system when it fights off bacteria and other invaders in injuries. Located around wounds , these chemicals cause pain and inflammation. Following bacterial infection, prostaglandins are also produced the hypothalamus, the brain’s center for controlling body temperature, resulting in a rise in temperature. In their capacities to cause pain, inflammation, and fever, prostaglandins are nuisances. Inhibiting their production, consequently reducing pain, inflammation, and fever, is the main therapeutic value of aspirin.
On the other hand, prostaglandins secreted by the stomach regulate acid production and maintain the mucus lining that protects the stomach from digesting itself. Prostaglandins in the blood’s platelets cause the platelets to stick together to initiate blood clotting in wounds. In these capacities, prostaglandins are crucial to a healthy body. Inhibiting their production leads to aspirin’s undesirable side effects, including upset stomach and excessive bleeding.
How does aspirin curb prostaglandin production? The many kinds of prostaglandin are synthesized by a host of complicated biochemical pathways. However, all pathways share a common stage facilitated by an enzyme called COX, whose action aspirin suppresses.
Enzymes are protein catalysts that speed up chemical reactions without being themselves used up in the reactions. An enzyme is a huge molecule with an active area that works somehow like a mold that accepts certain raw pieces and casts them into a final form. Imagine a mold that stamps a rod and a bowl into a spoon. Spoon production would be disrupted if someone throws a monkey range into the mold. Such a monkey range – an enzyme inhibitor – would make a desirable drug if it stops an enzyme from producing disease-inducing chemicals. Aspirin is an enzyme inhibitor. It suppresses the action of the enzyme COX, stops the production of prostaglandin, thus disrupting the pathways to pain, inflammation, elevated temperature, and stomach protection.
Vane’s success attracted many researchers to the area. Their investigations spread from aspirin to similar drugs that suppress pain and inflammation. By 1974, it was fairly well established that all NSAIDs act with similar mechanisms. They are all COX inhibitors.
Aspirin, ibuprofen, naproxen, and many other non-steroidal anti-inflammatory drugs (NSAIDs) work as COX inhibitors. They suppress the catalytic functions of the enzymes COX1 and COX2. COX2, which appears up injuries and other inflammatory stimuli, is deemed “bad”. It catalyzes the synthesis of prostaglandins that, located near sites of injuries, cause pain and inflammation. Inhibition of COX2 is responsible for the therapeutic effects of reducing pain, inflammation, and fever. COX1, which is present in many parts of the body, is deemed “good.” It catalyzes the synthesis of prostaglandins that perform many physiological functions, e.g., maintaining the mucus lining of the stomach or causing platelets in the blood to stick and form clots over wounds. Inhibition of COX1 is responsible for the drugs’ side effect of stomach irritation. In reducing the risk of blood clots, it is also responsible for aspirin’s efficacy in heart attack prevention. A new class of NSAID, COX2 inhibitor, is designed to target bad COX2 selectively and leave good COX1 alone, thus reducing pain and inflammation without upsetting the stomach.
From empiricism to concept-directed research
Connecting the dots has become a familiar phrase in the aftermath of September 11, 2001, when the intelligence community was blamed for failing to connect the dots and read from available information the message that terrorists were preparing to attack America. Dots can be connected arbitrarily to suit any agenda, but that is not what people want. They demand connections that reveal significant objective patterns.
Empirical observations yield dots of data. To connect them objectively depends on appropriate concepts and theories. The concept of COX-inhibition connects aspirin’s medicinal effects to prostaglandin and COX action. Due to the conceptual connection, these hitherto disparate pieces of knowledge become nexus in a network of relations. Furthermore, the network reveals other nexus and patterns that no one had dreamed of before. Therein lies the power of scientific theories to predict new phenomena, raise new questions, and indicate new areas for inquiry. That is why scientists highly value concepts and theories that correctly extract a set of relevant data and reveal their connections.
With the concept of COX-inhibition, knowledge about aspirin changes from mere empiricism to theory guided research. Whereas an empirical fact is specific to a particular phenomenon, a concept is general and potentially applicable to other phenomena. COX enzyme is present in many parts of the body, including unexpected places such as colon tumors. The conceptual framework of COX inhibition suggests links between aspirin and phenomena hitherto deemed unrelated, thus enabling scientists to ask significant questions and direct their research efforts. Basic scientists can use NSAIDs as tools to probe the physiological effects of COX, for instance in the formation of cancer.[xxi] Pharmaceutical firms can use COX enzyme in test tubes to screen for promising drugs. A conceptual framework that explains phenomena by their underlying mechanisms is not a last word but a scientific breakthrough. A final word closes the door on exploration, a breakthrough opens up a frontier of research.
Biochemistry meets molecular biology
The COX inhibition concept brought out many interesting questions.[xxii] The NSAIDs are similar but not identical. They exhibit considerable variations in their effects. For instance, ibuprofen is easer on the stomach than aspirin. How can the variations be explained in terms of COX inhibition? This calls on basic science to uncover the detailed mechanisms by which the drugs interact with COX enzyme.
Detailed mechanisms depend on the internal structure of COX, which biochemists did not know. They investigated external properties of COX and discovered that it behaves differently in different situations. Some COX enzymes, for instance those in the stomach, are present all the time. Others, for instance those responsible for pain-inducing prostaglandins, appear only at the heel of physiological injuries. This and other experimental results prompted biochemists to speculate that COX existed in two similar but distinct forms.
Further scientific research proceeded slowly. Even if two forms of COX existed, their catalytic actions were so similar it was difficult to tease them apart by biochemical means. The secret lies in the internal structure of COX, but this is a hard nut to crack. Unlike most drugs, which are rather simple molecules with tens or hundreds of atoms, COX and other enzymes are huge molecules with thousands of atoms and monstrous complexity.
So far, aspirin research mainly marched from the top down, from organisms to tissues to enzymes. Now biochemistry rendezvoused with a branch of science marching from the bottom up, from atoms to enzymes. When two sciences meet, their concepts mesh and many more dots are connected to yield improved intelligence. Using X-ray crystallography and other technologies, molecular biologists were unraveling the molecular structures of genes and the enzymes encoded by the genes. They discovered in 1991 a novel gene that coded for an enzyme highly similar, but not identical, to the COX that was isolated from smooth muscles and widely studied. Soon molecular biologists established that COX enzyme has two forms, called COX1 and COX2. The two are coded by different genes and serve different physiological functions.[xxiii]
Briefly, molecules of COX1 are widely distributed in the body’s cells, from the stomach to the platelets of the blood. Continuously present in the body, they serve “housekeeping” functions that maintain various normal physiological conditions. For its maintenance of healthy physiology, COX1 is duped “good.” Molecules of COX2 are not present all the time. Their production is induced by inflammatory and other injurious stimuli, and they tend to localize in the sites of injury, for instance the swelled joints of people suffering from rheumatoid arthritis. For its culpability in pain and inflammation, COX2 is duped “bad.”
Old COX inhibitors – NSAIDs such as aspirin and ibuprofen – inhibit the actions of both COX1 and COX2. Suppressing the bad COX2 accounts for the drugs’ therapeutic effects. Suppressing the good COX1 leads to their undesirable side effects. Different drugs have different selectivity for the two COXs, which partly explains their varying medicinal profiles. Unfortunately, they all tend to be harsher on the good COX1. The worse is aspirin itself. It is 150 times more effective in inhibiting COX1 than COX2, and is harsher on the stomach than its cousins.
Success and setback of rational drug design
The physiology of COX enzymes is complex. However, once the big picture of the COX1/COX2 concept is established, it does not take much for pharmacologists to recognize the appeal to design a drug that selectively inhibits the bad COX2 while sparing the good COX1.
Rational drug design is a very different approach than the trial and error that led to the first use of aspirin. To design drugs that target a specific enzyme or biochemical mechanism is increasingly feasible since the 1980s, when scientific knowledge about relevant mechanisms began to accumulate. Molecular biologists had deciphered the molecular sequences of the COX enzymes and how they fold into complex three-dimensional structures. Armed with this knowledge, pharmacologists set about finding small drug molecules that interfere with only COX2 and not COX1.[xxiv]
First generation COX2 inhibitors, Celebrex and Vioxx, reached consumers in 1999. Nicknamed “super aspirins,” they are comparable to aspirin in reducing pain and inflammation. Large scale clinical trials also found that they cause significantly less gastrointestinal irritation than the old COX inhibitors. Gastrointestinal side effects of COX inhibitors were blamed for roughly 100,000 hospitalizations and 15,000 deaths each year in the United States alone. Rheumatoid arthritic patients who had to take high dosages for long periods suffered most. To them COX2 inhibitors that promise to lessen the toll were godsend.[xxv]
Both Celebrex and Vioxx made the 2001 list of top ten drugs by global sales, beating the famous impotence drug Viagra, introduced in 1998. Clinical superiority is not the only reason for the high dollar sales of COX2 inhibitors. Another reason is that super aspirins are much more expensive than aspirin. Social critics grumble that the new drugs are not worth their high prices, but people whose drug bills are covered by health insurance do not care.[xxvi] They demand the best, whether or not aspirin upsets their stomachs; they pay the same insurance premium anyway. COX2 inhibitors grossed $5.7 billions and accounted for 23 percent of the pain and inflammation drug market in 2001. That was smaller than the 30 percent for the old COX inhibitors, but the trend of replacement was unmistakable. Optimistic analysts projected that by 2010, the market share of COX2 inhibitors would more than double that of COX inhibitors.[xxvii]
Optimism turned sour when the rising star of COX2 inhibitors took a major hit. Drugs on market are already approved by the Food and Drug Administration and not required to undergo further safety tests. Nevertheless, people are free to test them, perhaps in search for possible additional applications. Following indications that COX2 inhibitors may be effective in reducing the risks of colon cancer and Alzheimer’s disease, drug makers and independent research institutes conduct long-term clinical trials on them. In late 2004, a major trial found that almost twice the subjects who took Vioxx for eighteen months had strokes or heart attacks than subjects who took sugar pills. The trial was terminated midway. Vioxx was immediately withdrawn from the market. The entire class of COX2 inhibitors was in trouble. Nor were the old COX inhibitors spared. Other trials cast clouds over Celebrex and naproxen (Aleve, Naprosyn). The FDA came under intense fire for failure to monitoring long-term safety.[xxviii]
The saga of COX inhibitors continues. Already the second generation of COX2 inhibitors is in the development pipeline. As long as people need pain killers without side effects, research continues.
Aspirin and the heart – scientific results are inconclusive
The fall of COX2 inhibitors reveals the complexity of physiology. Their fault for raising risks for heart attacks does not come as a bolt from the blue. Years before they hit the market, Vane predicted that COX2 inhibitors would replace COX inhibitors in most areas except one: prevention of heart attacks.
COX1, which super aspirins spare but aspirin inhibits, is good but not absolutely good – nothing is absolute in a complex world. COX1 is indispensable in many healthy functions. Among them is blood clotting. If blood fails to clot and close a wound, one can bleed to death from a small cut, a danger well know to people suffering from hemophilia. Therefore the action of COX1 in blood clotting is crucial for most ordinary situations. However, in some rare cases, unwanted blood clot in the wrong places can also lead to heart attacks. In those heart-related circumstances, the action of aspirin as COX1 inhibitor would be beneficial.
Aspirin began its affair with the heart even before it got its name. A major reason that Bayer initially hesitated to market it was its perceived “enfeebling action on the heart.” That misperception persisted for decades, so much so that some aspirin advertisements carried the claim: “Does not affect the heart.” Judging the claim unsubstantiated, the U.S. government banned it in the 1930s. Fifty years later, government regulators faced the opposite claim: Aspirin does affect the heart – in a good way.[xxix]
Heart attack, the biggest killer in America, involves many factors. One is the narrowing of arteries by plaque such as cholesterol. Another is the formation of blood clots in arteries. If a blood clot blocks a narrowed artery, it can stop blood flow to the heart, resulting in damages of heart tissues, commonly called a heart attack. Or it can stop blood flow to the brain, resulting in a stroke. In the 1960s, some physicians hoped to reduce the risk of artery blood clots by blood thinning drugs. The hope collapsed; these drugs did seemed to reduce heart attacks, but they also increased fatal bleeding in the brain – a devil’s trade.[xxx]
Blood clotting is a complex process. The blood contains, besides red and white blood cells, partial cells called platelets. The disc-like platelets are produced in the bone marrow and cannot reproduce themselves because they contain no nucleus. They usually lie dormant in the blood, awakened only by chemicals released by injured tissues or a tear in the artery’s plaque. These stimulants activate the COX1 enzyme in the platelets to produce a prostaglandin, which causes the platelets to stick together, triggering the cascade of reactions that result in clotting of blood. By inhibiting COX1 from synthesizing the prostaglandin, aspirin reduces the stickiness of platelets, hence the chance of forming blood clots. For this antiplatelet purpose aspirin is uniquely effective. All other aspirin-like drugs inhibit COX temporarily, aspirin alone inhibits it permanently. One dose of aspirin has antiplatelet effects that last through the platelet’s lifetime, about ten days.
Aspirin’s antiplatelet effect was observed in 1967. Harvey Weiss and Louis Aledort divided their experimental subjects into two groups, gave aspirin to one but not the other. They then measured how long the subjects bleed from pinpricks and correlated it to the degree of platelet aggregation in blood taken from the subjects. Data revealed that those who had taken aspirin bleed longer and the platelets in their blood aggregated less. Weiss and Aledort suggested that aspirin may prevent artery blood clots.[xxxi] They did not know how aspirin prevented platelet aggregation, but that was explained four years later when Vane discovered aspirin’s COX inhibition effect.
Basic science and knowledge about underlying mechanisms strengthen the case for “an aspirin a day keeps heart attacks away.” However, they are not sufficient to prove it. We saw earlier that tissue bioassay is better than whole animal experiments in isolating a process and uncovering its underlying mechanism, which is buried under myriad processes going on in a life animal. The advantage in discovery can become a disadvantage in applying its results. In isolating a process we ignore its interaction with other processes in the context of application. These interactions can generate side effects or even derail the process itself. In the test tube, aspirin inhibits COX1 in platelets and hence the formation of a prostaglandin that promotes blood clots; fine. In the body, the situation is far more complex. For instance, aspirin also inhibits COX2 in blood vessels and hence the formation of another prostaglandin that prevents blood clots.[xxxii] How would the two processes of opposing effects balance out? Another question, would taking aspirin years on end, even at low dosages, increase the risk of bleeding in the brain? These and many other questions involving the functioning of the body as a whole cannot be answered by test-tube experiments on individual processes. That is why governments require drugs to pass clinical trials in human subjects to prove their effectiveness and safety.
Encouraged by the experimental results of Weiss and Aledort, epidemiologist Peter Elwood initiated the first clinical trial of aspirin’s efficacy in preventing second heart attacks in 1971. It ended thirty months later with the all-too familiar remark: “The results of this trial were inconclusive.”[xxxiii]
Elwood persisted with further trials, so did other academicians and pharmaceutical companies. A three-year and $17 million experiment funded by the U.S. government returned in 1980 the disheartening result that aspirin had no effect on the heart. However, other trials returned mildly favorable results. Scientific debates ensued. Trial protocols were criticized, new experiments designed and launched, data analyzed and reanalyzed and aggregated with sophisticated mathematics. Finally in 1985, the U.S. Food and Drug Administration approved the claim that long-term low-dosage aspirin reduces the risk for recurrence of heart attack.[xxxiv]
Trials on people with other health conditions continue. Aspirin taken by patients while having a heart attack significantly reduces the chance of death. For people with various risk factors for heart diseases such as diabetes, obesity, or high blood pressure, aspirin lowers the probability of a first heart attack. For healthy people without cardiovascular risks, results on the effects of aspirin are inconclusive.[xxxv]
Chances are aspirin will make news again. Its continuing story celebrates the persistent advancement of science. [i]. G. Weissman. Aspirin. Scientific American, 264(1): 84-90 (1991). D. B. Jack. A hundred years of aspirin. The Lancet, 350: 437-439 (1997). [ii]. V. Fuster, M. Cohen, and J. Halperin. Aspirin in the prevention of coronary disease. New England Journal of Medicine, 321: 183-185 (1989). M. J. Underwood. The aspirin papers. British Medical Journal: 308: 71-72 (1994). PPP (Collaborative Group of the Primary Prevention Project). H. J. M. Barnett, M. Eliasziw, and H. E. Meldrum. Prevention of ischaemic stroke. British Medical Journal, 318, 1539-1543 (1998). Low-does aspirin and vitamin E in people at cardiovascular risk: a randomized trial in general practice. The Lancet, 357: 89-95 (2001). [iii]. S. J. Shiff and B. Rigas. Aspirin for cancer. Nature Medicine, 5: 1348-1349 (1999). T. F. Imperiale. Aspirin and the prevention of colorectal cancer. The New England Journal of Medicine, 348: 879-880 (2003). D. N. DuBois. Aspirin and breast cancer prevention: the estrogen connection. Journal of the American Medical Association, 291: 2488-2489 (2004). [iv]. D. Lawrence. Retrospective data strengthen Alzheimer’s link with aspirin and NSAIDs. The Lancet, 360: 1003 (2002). [v]. Acetaminophen (Tylanol) is not a NSAID, because it does not suppress inflammation. [vi]. J. R. Vane. Towards a better aspirin. Nature, 367: 215-216 (1994). E. Pennisi. Building a better aspirin. Science, 280: 1191-1192 (1998). [vii]. D. Jeffreys. Aspirin, The Remarkable Story of a Wonder Drug, New York: Bloomsbury (2005). R. Porter, R. 1997. The Greatest Benefit to Mankind. New York: Norton (1997), p. 448. J. Drews. In Quest of Tomorrow’s Medicines. New York: Springer (1999), pp. 43-44. [viii]. Drews, Quest, pp. 24-26. B. Achilladelis. Innovations in the pharmaceutical industry. In Pharmaceutical Innovations, eds. R. Landau, B. Achilladelis, and A. Scriabine, Philadelphia: Chemical Heritage Press (1999). pp. 1-147. [ix]. J. J. Beer. Coal tar dye manufacture and the origin of modern industrial research laboratory. Isis 49, part 2 (156): 123-31 (1958). E. Homburg. The emergence of research laboratories in the dyestuffs industry, 1870-1900. British Journal of History of Science, 25: 91-111 (1992). [x]. P. Collard. The Development of Microbiology. New York: Cambridge University Press (1976), ch. 5. [xi]. C. C. Mann and M. L. Pulmmer. The Aspirin Wars. New York: Knopf (1991), pp. 23-26. [xii]. Achilladelis, Innovations. [xiii]. G. Dutfield, Intellectual Property Rights and the Life Science Industries, Aldershot, UK: Ashgate (2003), ch. 2. [xiv]. The trade mark ASPIRIN still applies in Germany, although elsewhere in the world aspirin becomes a generic name. [xv]. Mann and Pulmmer. The Aspirin Wars, pp. 185-192. [xvi]. H. Henderson. The paternity of aspirin. Science, 286: 1089 (1999). [xvii]. S. Chrubasik, et al. Treatment of low back pain exacerbations with willow bark extract: a randomized double-blind study. American Journal of Medicine, 109: 9-14 (2000). [xviii]. Mann and Plummer, Aspirin Wars, p. 273. [xix]. Mann and Plummer, Aspirin Wars, ch. 17. J. R. Vane. History of aspirin and its mechanism of action. Stroke, 21 (supplement 4): 12-23 (1990). R. J. Flower. The development of COX2 inhibitors. Nature Review: Drug Discovery, 2: 179-191 (2003). [xx]. J. R. Vane. Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nature, New Biology: 231: 230-235 (1971). [xxi]. L. J. Marnett and R. N. DuBois. COX2: a target for colon cancer prevention. Annual Review of Pharmacology and Toxicology, 42: 55-80 (2002). [xxii]. Weissman, Aspirin. [xxiii]. Flower, COX2. [xxiv]. Flower, COX2. Vane, Better aspirin. [xxv]. Flower, COX2. Pinni, Better aspirin. J. R. Vane. Back to an aspirin a day? Science, 296: 747-745 (2002). [xxvi]. W. L. Peterson and B. Cryer. COX-1 sparing NSAIDS – is the enthusiasm justified? JAMA, 282: 1961-1963 (1999). C. A. Marra, et al. The cost of COX inhibitors: How selective should we be? Journal of Rheumatology, 27: 2731-2733 (2000). [xxvii]. S. Renfrey, C. Downton, and J. Featherstone. The painful reality. Nature Review: Drug Discovery, 2: 175-176 (2003). www.ims-global.com, accessed May, 2004. The other half of the market belongs to drugs for neuropathic pain, migraine, and opioids for sever pain. [xxviii]. J. Couzin. Withdrawal Vioxx casts a shadow over COX-2 inhibitors. Science, 306: 384-385 (2004). S. Frantz. Vioxx risk could signify trouble in class. Nature Reivews Drug Discovery, 3: 899-901 (2004). [xxix]. Vane, Better aspirin. Mann and Plummer, Aspirin Wars, pp. 25, plates after p. 214. [xxx]. G. Pickerings, et al. An assessment of long-term anticoagulant administration after cardiac infraction. British Medical Journal, October 3, pp. 837-843 (1964). [xxxi]. H. J. Weiss and L. M. Aledort. Impaired platelet/connective tissue reaction in man after aspirin injection. The Lancet, September 2: 495-497 (1967). [xxxii]. Vane, Back to aspirin. [xxxiii]. Mann and Plummer, Aspirin Wars, pp. 265-269. [xxxiv]. Mann and Plummer, Aspirin Wars, pp. 291-309. [xxxv]. See note 1.
|